WHAT IS CHLORINATION FACT SHEET
What is Chlorination?
Microorganisms can be found in raw water from rivers, lakes and groundwater. While not all microorganisms are harmful to human health, there are some that may cause diseases in humans. These are called pathogens. Pathogens present in water can be transmitted through a drinking water distribution system, causing waterborne disease in those who consume it.
In order to combat waterborne diseases, different disinfection methods are used to inactivate pathogens. Along with other water treatment processes such as coagulation, sedimentation, and filtration, chlorination creates water that is safe for public consumption.
Chlorination is one of many methods that can be used to disinfect water. This method was first used over a century ago, and is still used today. It is a chemical disinfection method that uses various types of chlorine or chlorine-containing substances for the oxidation and disinfection of what will be the potable water source.
The History of Chlorination
Chlorine was first discovered in Sweden in 1744. At that time, people believed that odours from the water were responsible for transmitting diseases. In 1835, chlorine was used to remove odours from the water, but it wasn't until 1890 that chlorine was found to be an effective tool for disinfecting; a way to reduce the amount of disease transmitted through water. With this new find, chlorination began in Great Britain and then expanded to the United States in 1908 and Canada by 1912. Today, chlorination is the most popular method of disinfection and is used for water treatment all over the world.
Why do we Chlorinate our water?
A large amount of research and many studies have been conducted to ensure success in new treatment plants using chlorine as a disinfectant. A leading advantage of chlorination is that it has proven effective against bacteria and viruses; however, it cannot inactivate all microbes. Some protozoan cysts are resistant to the effects of chlorine.
In cases where protozoan cysts are not a major concern, chlorination is a good disinfection method to use because it is inexpensive yet effective in disinfecting many other possibly present contaminants. The chlorination process is also fairly easy to implement, when compared to other water treatment methods. It is an effective method in water emergency situations as it can eliminate an overload of pathogens relatively quickly. An emergency water situation can be anything from a filter breakdown to a mixing of treated and raw water.
How does Chlorine Inactivate Microorganisms?
Chlorine inactivates a microorganism by damaging its cell membrane. Once the cell membrane is weakened, the chlorine can enter the cell and disrupt cell respiration and DNA activity (two processes that are necessary for cell survival).
When/How do We Chlorinate Our Waters?
Chlorination can be done at any time/point throughout the water treatment process - there is not one specific time when chlorine must be added. Each point of chlorine application will subsequently control a different water contaminant concern, thus offering a complete spectrum of treatment from the time the water enters the treatment facility to the time it leaves.
Pre-chlorination is when chlorine is applied to the water almost immediately after it enters the treatment facility. In the pre-chlorination step, the chlorine is usually added directly to the raw water (the untreated water entering the treatment facility), or added in the flash mixer (a mixing machine that ensures quick, uniform dispersion of the chlorine). Chlorine is added to raw water to eliminate algae and other forms of aquatic life from the water so they won’t cause problems in the later stages of water treatment. Pre-chlorination in the flash mixer is found to remove tastes and odours, and control biological growth throughout the water treatment system, thus preventing growth in the sedimentation tanks (where solids are removed from the water by gravity settling) and the filtration media (the filters through which the water passes after sitting in the sedimentation tanks). The addition of chlorine will also oxidize any iron, manganese and/or hydrogen sulphide that are present, so that they too can be removed in the sedimentation and filtration steps.
Disinfection can also be done just prior to filtration and after sedimentation. This would control the biological growth, remove iron and manganese, remove taste and odours, control algae growth, and remove the colour from the water. This will not decrease the amount of biological growth in the sedimentation cells.
Chlorination may also be done as the final step in the treatment process, which is when it is usually done in most treatment plants. The main objective of this chlorine addition is to disinfect the water and maintain chlorine residuals that will remain in the water as it travels through the distribution system. Chlorinating filtered water is more economical because a lower CT value is required. This is a combination of the concentration (C) and contact time (T). The CT concept is discussed later on in this fact sheet. By the time the water has been through sedimentation and filtration, a lot of the unwanted organisms have been removed, and as a result, less chlorine and a shorter contact time is required to achieve the same effectiveness. To support and maintain the chlorine residual, a process called re-chlorination is sometimes done within the distribution system. This is done to ensure proper chlorine residual levels are maintained throughout the distribution system.
Residual Chlorine, Breakpoint
Any type of chlorine that is added to water during the treatment process will result in the formation of hypochlorous acid (HOCl) and hypochlorite ions (OCl-), which are the main disinfecting compounds in chlorinated water. More detail is provided later on in this fact sheet.
A Form of Chlorine + H2O -> HOCl + OCl-
Of the two, hypochlorous acid is the most effective. The amount of each compound present in the water is dependent on the pH level of the water prior to addition of chlorine. At lower pH levels, the hypochlorous acid will dominate. The combination of hypochlorous acid and hypochlorite ions makes up what is called ‘free chorine.’ Free chlorine has a high oxidation potential and is a more effective disinfectant than other forms of chlorine, such as chloramines. Oxidation potential is a measure of how readily a compound will react with another. A high oxidation potential means many different compounds are able to react with the compound. It also means that the compound will be readily available to react with others.
Combined chlorine is the combination of organic nitrogen compounds and chloramines, which are produced as a result of the reaction between chlorine and ammonia. Chloramines are not as effective at disinfecting water as free chlorine due to a lower oxidation potential. Due to the creation of chloramines instead of free chlorine, ammonia is not desired product in the water treatment process in the beginning, but may be added at the end of treatment to create chloramines as a secondary disinfectant, which remains in the system longer than chlorine, ensuring clean drinking water throughout the distribution system.
The amount of chlorine that is required to disinfect water is dependent on the impurities in the water that needs to be treated. Many impurities in the water require a large amount of chlorine to react with all the impurities present. The chlorine added must first react with all the impurities in the water before a chlorine residual is present. The amount of chlorine that is required to satisfy all the impurities is termed the ‘chlorine demand.’ This can also be thought of as the amount of chlorine needed before free chlorine can be produced. Once the chlorine demand has been met, breakpoint chlorination (the addition of chlorine to water until the chlorine demand has been satisfied) has occurred. After the breakpoint, any additional chlorine added will result in a free chlorine residual proportional to the amount of chlorine added. Residual chlorine is the difference between the amount of chlorine added and the chlorine demand. Most water treatment plants will add chlorine beyond the breakpoint.
If ammonium is present in the water at the time of chlorine addition breakpoint chlorination will not occur until all the ammonium has reacted with the chlorine. Between 10 and 15 times more chlorine than ammonia is required before free chlorine and breakpoint chlorination can be achieved. Small water treatment plants frequently only add a fraction of the required chlorine (in relation to ammonium ions) and end up not properly disinfecting their water supplies.
The type of chloramines that are formed is dependent on the pH of the water prior to the addition of chlorine. Between the pH levels 4.5 and 8.5, both monochloramine and dichloramine are created in the water. At a pH of 4.5, dichloramine is the dominant form, and below that trichloramine dominates. At a pH above 8.5 monochloramine is the dominant form. Hypochlorous acid reacts with ammonia at its most rapid rate at a pH level around 8.3.
The chlorine to ammonia nitrogen ratio characterizes what kind of residual is produced.
Are there Other Uses for Chlorine?
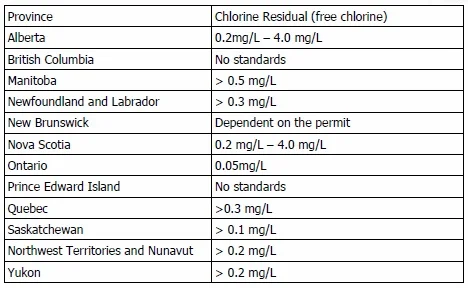
The main purpose of chlorination is to disinfect water, but it also has many other benefits. Unlike some of the other disinfection methods like ozonation and ultraviolet radiation, chlorination is able to provide a residual to reduce the chance of pathogen regrowth in water storage tanks or within the water distribution system. At times, distribution systems can be a fair distance from the storage tanks and in dead end sections or where water is not used pathogens may re-grow if a proper (chlorine) residual is cannot be maintained in the treated water sent out for consumption. This results in poor water quality as well as slime and biofilms in the distribution systems that will end up contaminating the clean, treated water being distributed. Many government environmental bodies have set guidelines or standards for the amount of chlorine residual that must be present at all points in the system. The guidelines for each province are shown in the table below.
In addition to providing a residual, adding chlorine to water will also: oxidize iron, manganese, taste and odour compounds, remove colour in the water, destroy hydrogen sulphide, and aid other water treatment processes, such as sedimentation and filtration. Oxidizing soluble reduced iron and manganese will result in particle formation as oxidized iron and manganese are not soluble in water.
Is Chlorine All the Same?
The chlorination process involves adding chlorine to water, but the chlorinating product does not necessarily have to be pure chlorine. Chlorination can also be carried out using chlorine-containing substances. Depending on the pH conditions required and the available storage options, different chlorine-containing substances can be used. The three most common types of chlorine used in water treatment are: chlorine gas, sodium hypochlorite, and calcium hypochlorite.
Chlorine Gas
Chlorine gas is greenish yellow in colour and very toxic. It is heavier than air and will therefore sink to the ground if released from its container. It is the toxic effect of chlorine gas that makes it a good disinfectant, but it is toxic to more than just waterborne pathogens; it is also toxic to humans. It is a respiratory irritant and it can also irritate skin and mucus membranes. Exposure to high volumes of chlorine gas fumes can cause serious health problems, including death. However, it is important to realize that chlorine gas, once entering the water, changes into hypochlorous acid and hypochlorite ions, and therefore its human toxic properties are not found in the drinking water we consume.
Chlorine gas is sold as a compressed liquid, which is amber in color. Chlorine, as a liquid, is heavier (more dense) than water. If the chlorine liquid is released from its container it will quickly return back to its gas state. Chlorine gas is the least expensive form of chlorine to use. The typical amount of chlorine gas required for water treatment is 1-16 mg/L of water. Different amounts of chlorine gas are used depending on the quality of water that needs to be treated. If the water quality is poor, a higher concentration of chlorine gas will be required to disinfect the water if the contact time cannot be increased.
When chlorine gas (Cl2) is added to the water (H2O), it hydrolyzes rapidly to produce hypochlorous acid (HOCl) and the hypochlorous acid will then dissociate into hypochlorite ions (OCl-) and hydrogen ions (H+).
Cl2 + H2O -> HOCl + H+ + OCl-
Because hydrogen ions are produced, the water will become more acidic (the pH of the water will decrease). The amount of dissociation depends on the original pH of the water. If the pH of the water is below a 6.5, nearly no dissociation will occur and the hypochlorous acid will dominate. A pH above 8.5 will see a complete dissociation of chlorine, and hypochlorite ions will dominate. A pH between 6.5 and 8.5 will see both hypochlorous acid and hypochlorite ions present in the water. Together, the hypochlorous acid and the hypochlorite ions are referred to as free chlorine. Hypchlorous acid is the more effective disinfectant, and therefore, a lower pH is preferred for disinfection.
Calcium Hypochlorite
Calcium hypochlorite (CaOCl) is made up of the calcium salts of hypochlorous acid. It is produced by dissolving chlorine gas (Cl2) into a solution of calcium oxide (CaO) and sodium hydroxide (NaOH). Calcium hypochlorite is a white, corrosive solid that comes either in tablet form or as a granular powder. Calcium hypochlorite is very stable, and when packaged properly, large amounts can be purchased and stored until needed. The chemical is very corrosive however, and thus requires proper handling when being used to treat water. Calcium hypochlorite needs to be stored in a dry area and kept away from organic materials. It cannot be stored near wood, cloth or petrol because the combination of calcium hypochlorite and organic material can create enough heat for an explosion. It must also be kept away from moisture because the tablets/granular powder readily adsorb moisture and will form (toxic) chlorine gas as a result. Calcium hypochlorite has a very strong chlorine odour – something that should be kept in mind when placing them in storage.
When treating water, a lesser amount of calcium hypochlorite is needed than if using chlorine gas. Compared to the 1-16 mg/L required with chlorine gas, only 0.5-5 mg/L of calcium hypochlorite is required. When calcium hypochlorite is added to water, hypochlorite and calcium ions are produced.
Ca(OCl)2 -> Ca+2 + 2OCl-
Instead of decreasing the pH like chlorine gas does, calcium hypochlorite increases the pH of the water (making the water less acidic). However, hypochlorous acid and hypochlorite concentrations are still dependent on the pH of the water; therefore by decreasing the pH of the water, hypochlorous acid will still be present in the water. As a result, calcium hypochlorite and chlorine gas both produce the same type of residuals.
Sodium Hypochlorite
Sodium hypochlorite (NaOCl) is made up of the sodium salts of hypochlorous acid and is a chlorine-containing compound that can be used as a disinfectant. It is produced when chlorine gas is dissolved into a sodium hydroxide solution. It is in liquid form, clear with a light yellow color, and has a strong chlorine smell. Sodium hypochlorite is extremely corrosive and must be stored in a cool, dark, and dry place. Sodium hypochlorite will naturally decompose; therefore it cannot be stored for more than one month at a time. Of all the different types of chlorine available for use, this is the easiest to handle.
The amount of sodium hypochlorite required for water treatment is much less than the other two forms of chlorine, with 0.2-2 mg of NaOCl/L of water being recommended. Like calcium hypochlorite, sodium hypochlorite will also produce a hypochlorite ion, but instead of calcium ions, sodium ions are produced. NaOCl will also increase the pH of the water through the formation of hypochlorite ions. To obtain hypochlorous acid, which is a more effective disinfectant, the pH of the water should be decreased.
NaOCl -> Na+ + OCl-
Is Chlorine a Sure Way of Eliminating Pathogens?
Chlorination has been proven to be very effective against bacteria and viruses. However, it cannot disinfect all waterborne pathogens. Certain pathogens, namely protozoan cysts, are resistant to the effects of chlorine. Cryptosporidium and Giardia, two examples of protozoan cysts, have caused great concern due to the serious illnesses they can cause. Cryptosporidium was the cause of the outbreak in North Battleford in 2001, and Milwaukee in April 1993. In raw water with high Giardia and Cryptosporidium levels, another method of disinfection should be considered. For more information on these protozoa, please read their self-titled fact sheets in the public information section.
Is Chlorinating Water ‘Fool-proof’?
There are a number of factors that affect the disinfection process. Of these, the concentration or dosage of chlorine and the chlorine contact time (the time that chlorine is allowed to react with any impurities in the water) are the most important factors.
Chlorine needs time to inactivate any microorganisms that may be present in the water being treated for human consumption. The more time chlorine is in contact with the microorganisms, the more effective the process will be. The contact time is the time from when the chlorine is first added until the time that the water is used or consumed.
The same positive relationship is seen when considering the chlorine concentration. The higher the concentration of chlorine, the more effective the water disinfection process will be. This relationship holds true because as the concentration increases, the amount of chlorine for disinfection is increased. Unlike the relationship between chlorine concentration and disinfection effectiveness, the chlorine concentration and the contact time of chlorine with water show an inverse relationship. As the chlorine concentration increases, the required water-chlorine contact time ultimately decreases. To determine the level of disinfection (D), a CT value can be calculated. This value is the product of the chlorine concentration (C) and contact time (T). The formula is as follows: C*T=D. This concept shows that an increase in chlorine concentration (C) would require less contact time to achieve the same desired level of disinfection. Another possibility would be an increase in contact time that would in turn require a lower chlorine concentration in order for the level of disinfection to stay the same.
The required CT value depends on several factors, including: the type of pathogens in the water, the turbidity of the water, the pH of the water and the temperature of the water. Turbidity is the suspended matter in the water and the types of pathogens can range from bacteria like E.coli and Campylobacter to viruses including Hepatitis A. At lower temperatures, higher turbidity, or higher pH levels, the CT value (i.e. the disinfection level) will have to be increased, but at lower turbidity, there is less suspended material in the water that will prevent contact of the disinfectant with the microorganisms, thus requiring a lower CT value. A higher water temperature and a lower pH level will also allow for a lower CT value.
Impurity Reactions
Chlorine can react with a number of different substances. In raw water, there may be a number of different impurities to react with the added chlorine, resulting in an increase of the chlorine demand. As a result, more chlorine will need to be added for the same level of inactivation. Some major impurities that may exist in water include: dissolved iron, hydrogen sulphide, bromine, ammonia, nitrogen dioxide, and organic material. In some cases, the result of chlorine reacting with impurities will increase the quality of the water (by eliminating the undesired elements), while in other cases, the chlorine-impurity reactions will create undesired side products that are harmful to human health. Chlorine will first react with inorganic impurities (dissolved iron, bromine, ammonia, etc.) before reacting with the organic compounds (dissolved organic material, bacteria, viruses, etc.).
Iron, which will give water an undesirable metallic taste if present, is one of the inorganic compounds that will react with hypochlorous acid (the stronger form of free chlorine that is produced after pure chlorine is added to water). By reacting with hypochlorous acid, the dissolved iron will go from a soluble state to an insoluble state, as a precipitate is formed as a result of the reaction. The iron precipitate, in its insoluble state, can be removed by filtration process within the water treatment centre.
2 Fe2+ (liquid) + HOCl + 5H2O -> 2 Fe(OH)3 (solid) + 5H+ + Cl-
Hypochlorous acid can also react with hydrogen sulphide (H2S), if it is present in the water being treated. Hydrogen sulfide is an undesirable impurity in water because it gives water an undesired smell. At levels below 1 mg/L hydrogen sulphide generates a musty smell to the water, while at levels above 1 mg/L a rotten egg smell will prevail. Hydrogen sulphide is also toxic. The hypochlorous acid and H2S reaction gives hydrochloric acid and sulphur ions as its products.
H2S + HOCl -> H+ + Cl- + S + H2O
Bromine in the water can result in the production of undesired compounds. Bromine ions can react with hypochlorous acid to create hypobromous acid. Hypobromous acid also has disinfectant properties and is more reactive than hypochlorous acid. Hypochlorous acid or hypobromous acid will react with organic material in the water and create halogenated by-products, such as trihalomethanes.
Br- + HOCl -> HOBr + Cl-
Ammonia is a compound that may exist in the water. It is a nutrient to aquatic life, but one that will become toxic in high concentrations. Ammonia is produced as a result of decaying matter and therefore naturally exists in the water; however, human activity also releases a large amount of ammonia into the water, which contributes to an increasing level of ammonia that may cause concern. Some ‘human activity sources’ include: municipal wastewater treatment plants, agricultural releases, and industrial releases, such as pulp and paper mills, mines, food processing, and fertilizer production. Reactions between ammonia and chlorine will produce monochloramines, dichloramines, and trichloramines, which are collectively known as chloramines. These compounds are beneficial to the water treatment process as they have disinfection capacity, but they are not as effective as chlorine although chloramines will last longer in the water.
Chlorine also reacts with phenols to produce monochlorophenols, dichlorophenols, or trichlorophenols, which cause taste and odour problem at low levels. At higher levels, chlorophenols are toxic and affect the respiration and energy storage process. Chlorophenols are mainly man-made compounds, but can be found naturally in animal wastes and decomposing organic material.
Are there Health Concerns with Chlorinating Water?
Chlorine can be toxic not only for microorganisms, but for humans as well. To humans,
chlorine is an irritant to the eyes, nasal passages and respiratory system. Chlorine gas must be carefully handled because it may cause acute health effects and can be fatal at concentrations as low as 1000 ppm. However, chlorine gas is also the least expensive form of chlorine for water treatment, which makes it an attractive choice regardless of the health threat.
In drinking water, the concentration of chlorine is usually very low and is thus not a concern in acute exposure. More of a concern is the long term risk of cancer due to chronic exposure to chlorinated water. This is mainly due to the trihalomethanes and other disinfection by-products, which are by-products of chlorination. Trihalomethanes are carcinogens, and have been the topic of concern in chlorinated drinking water. Chlorinated water has been associated with increased risk of bladder, colon and rectal cancer. In the case of bladder cancer, the risk may be doubled. Although there are concerns about carcinogens in drinking water, Health Canada's Laboratory Centre for Disease Control says that the benefits of chlorinated water in controlling infectious diseases outweigh the risks associated with chlorination and would not be enough to justify its discontinuation. In Europe, however, chorination has been discontinued in many communities.
Chlorination By-products
A number of different by-products can be produced from the reactions in the disinfection process. By-products created from the reactions between inorganic compounds and chlorine are harmless and can be easily removed from the water by filtration. Other by-products, such as chloramines, are beneficial to the disinfection process because they also have disinfecting properties. However, there are undesired compounds that may be produced from chlorine reacting with organic matter. The compounds of most concern right now are trihalomethanes (THMs) and haloacetic acids (HAAs). THMs and HAAs are formed by reactions between chlorine and organic material such as humic acids and fulvic acids (both generated from the decay of organic matter) to create halogenated organics. A greater level of THM formation has been found in surface water or groundwater influenced by surface water.
Trihalomethanes are associated with several types of cancer and are considered carcinogenic. The trihalomethane of most concern is chloroform, also called trichloromethane. It was once used as an anaesthetic during surgery, but is now used in the process of making other chemicals. About 900 ppm of chloroform can cause dizziness, fatigue, and headaches. Chronic exposure may cause damage to the liver and kidneys. Other harmful disinfection by-products are: trichloracetic acid, dichloroacetic acid, some haloacetonitriles, and chlorophenols.
Trichloracetic acid is produced commercially for use as a herbicide and is also produced in drinking water. This chemical is not classified as a carcinogen for humans, and there is limited information for animals. Dichloroacetic acid is an irritant, corrosive, and destructive against mucous membranes. This is also not currently classified as a human carcinogen. Haloacetonitriles were used as pesticides in the past, but are no longer manufactured. They are produced as a result of a reaction between chlorine, natural organic matter, and bromide. Chlorophenols cause taste and odour problems. They are toxic, and when present in higher concentrations, affect the respiration and energy storage process in the body.
Conclusion
Chlorination is a very popular method of water disinfection that has been used for many years. It has shown to be effective for killing bacteria and viruses, but not for some protozoan cysts. With the concern about trihalomethanes, a carcinogenic disinfection by-product, many communities have become hesitant in the continuation of this process.
Although chlorination does have some drawbacks, it continues to be the most popular, dependable, and cost-effective method of water disinfection.
Find this useful? Please chip in $5 to help us send Operation Water Drop kits to schools so students can measure the amount of total chlorine in the water they drink every day! Or donate $20 or more and receive an Official Donation Receipt for Income Tax Purposes.